A new technique could pave the way for producing uniform nanoparticles for use in drug delivery systems.
Scientists have been investigating how to make better use of nanoparticles in medicine for several decades. Significantly smaller than an average cell, nanoparticles are more similar in size to proteins. This makes them good at interacting with biomolecules and transporting drug molecules attached to their surface across cell membranes.
To date, however, only a handful of nanoparticle-based drugs have succeeded in reaching the clinic. This is because of the challenges in controlling the size and shape of nanoparticles – and understanding fully how these variables affect the way the particles behave in the body.
In a new study, published in Nature Communications, researchers from the University of Bath and the University of Birmingham have demonstrated a technique that will allow chemists to more closely control the size and shape of nanoparticles.
Dr Tom Wilks, in the University of Birmingham’s School of Chemistry, is one of the lead authors of the study. He explains: “If you change the shape of a nanoparticle from, for example, a spherical to a cylindrical shape, others have shown that this can have a dramatic effect on how it interacts with cells in the body, and how it is distributed through the body. By being able to control the size and shape, we can start to design and test nanoparticles that are exactly suited to an intended function.”
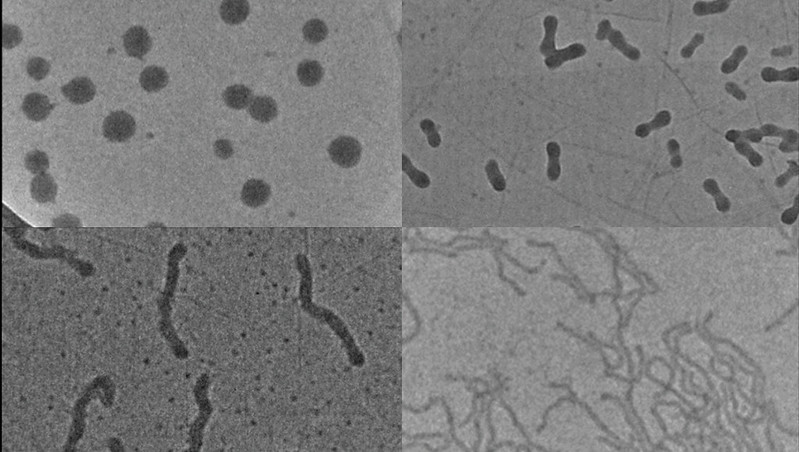
Dr Anton Souslov, from the University of Bath’s Department of Physics, said: “Rain drops tend to be spherical because of surface tension, and this effect is even more pronounced if the droplets are so tiny as to be nanoscale. However, for applications from drug delivery to fuel cells and the control of light, non-spherical particles can work better. We are excited about this research because it shows a new way of overcoming surface tension and producing elongated, worm-like nanoparticles. Starting with these shapes, we hope it will be possible to engineer the particles and tailor them to each specific application.”
Currently, to produce differently shaped nanoparticles for drug delivery scientists have to develop a bespoke chemical synthesis for each, which can be a laborious, time-consuming and expensive process.
The technique developed by the research team offers a deceptively simple way of streamlining this process. The team started with a base nanoparticle, made of a polymer, and added a second polymer in solution. The polymers are designed so they want to bond to each other, so the second polymer is driven into the core of the nanoparticle, forcing it to expand. The exact size and shape of the nanoparticle is then determined simply by how much of the second polymer is added.
“The precise way that these polymers were designed and the control we have over how much of the second polymer is added means we can accurately predict the shape of the nanoparticle, and have a high degree of control over its size,” explains Dr Wilks. The team believe the process could also be reproduced with other polymers, meaning the process could be adapted for any number of applications involving nanoparticles, from photonics to fuel cells.
“This is an important first step in being able to effectively harness nanoparticles for a whole host of applications, but there are a lot of questions still to answer,” says Dr Wilks. “For example, in the field of drug delivery, we need to know much more about what would happen once drug molecules are introduced to our nanoparticles, as well as how the sizes and shapes of the nanoparticles can be optimised for different uses.”