New research published today in the journal Proceedings of the National Academy of Sciences (PNAS) from researchers in our Department for Health with international colleagues has shown a biological clock at work in our muscle cells, which could be an important factor in regulating our metabolism and in the development of diabetes.
Biological clocks are ticking everywhere throughout our body. They trigger the release of the hormone melatonin during sleep, leading to the secretion of digestive enzymes at lunchtime or keep us awake at the busiest moments of the day. A "master clock" in the brain synchronises all the subsidiary ones in various organs.
For this study, researchers funded by the Swiss National Science Foundation (SNSF), found that such a circadian clock is also at work in our muscles. Such a finding could have important implications for healthcare and treatments. It is thought that this system going wrong could be an important factor in the development of type 2 diabetes.
Sampling muscle tissue every four hours
Researchers from our University, along with the University of Geneva, the Université Claude Bernard in Lyon, EPFL, the University of Surrey, and the Nestlé Institute of Health Sciences discovered that levels of the various types of fat (lipids) contained in our muscle cells vary during the day, sometimes favouring one kind of lipid over another.
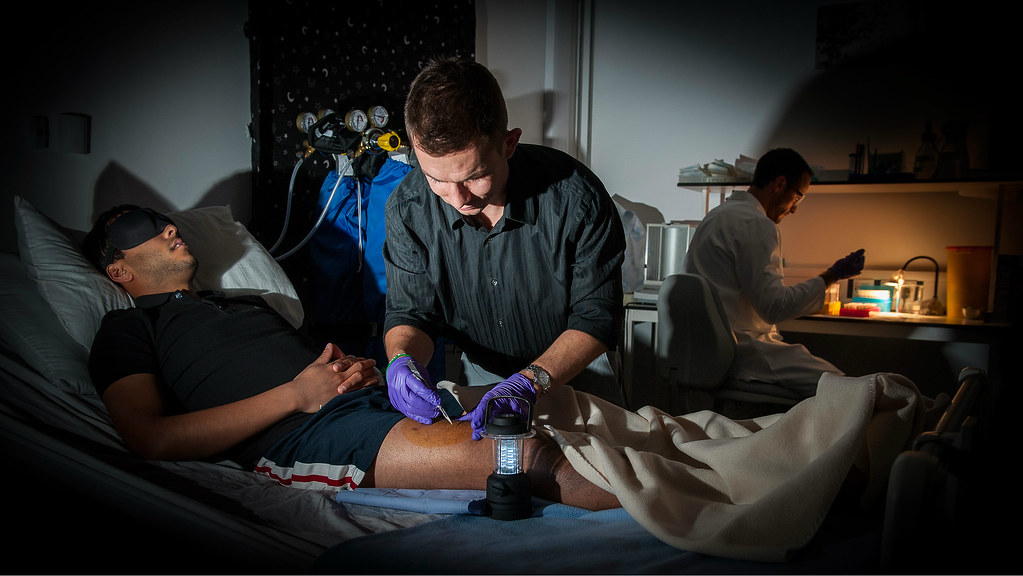
The international team has tested the hypothesis that a biological clock could be at play. Testing on volunteer subjects at the University of Bath, they synchronised every subject’s master clock by prescribing them identical light periods as well as meals and exercise time for up to two days. Every four hours, researchers took a very small sample of thigh muscle tissue and analysed its lipid composition.
For the study – the first of its kind - volunteers stayed in labs at the University of Bath for 37 hours consecutively. Throughout their stay they were kept in a rested state whilst receiving hourly feedings during waking hours to meet energy requirements, before sleeping from 10pm to 7am. Every four hours researchers took a very small sample of muscle tissue from the thigh and analysed its lipid composition to see how it varied over the course of a typical 24 hour period.
Dr Betts, who led the work at Bath, explains: “We realised we were ideally placed to sample serial muscle biopsies every 4 hours throughout the day and night, thus providing the first evidence of these important biological rhythms in human skeletal muscle.”
The team at Bath also involved Dr Jean-Philippe Walhin and Iain Templeman.
With the samples obtained, the team observed a clear correlation between the muscle cell’s lipid composition and the time of day, explains Howard Riezman, who co-directed the study in Geneva with colleague Charna Dibner.
He said: “As the combination of lipids varied substantially from one individual to another, we needed further evidence to corroborate these findings.”
In a second step, the researchers switched to an in-vitro experiment. They cultivated human muscle cells and artificially synchronised them in the absence of a master clock, using a signal molecule normally secreted in the body. This highlighted the levels of fat in the body – the cell’s lipid composition – varied at different points in the day. When they disrupted the clock mechanism by inhibiting the responsible genes, this variation in lipid composition was lost.
Diabetes and sleep disorders are linked
First author on the paper, Ursula Loizides-Mangold said: “We have clearly shown that this variation of lipid types in our muscles is due to our circadian rhythm. But the main question is still to be answered: what is this mechanism for?”
Riezman thinks that the biological clock in the muscle, with its impact on the lipids, could help in regulating the cells' sensitivity to insulin. Indeed, lipids – being a component of the cell membrane – influence the molecules' ability to travel into and out of the muscle cells. Changes in its composition could tune the muscle's sensitivity to the hormone as well as its ability to take in blood sugar.
A low sensitivity of the muscle to insulin leads to a condition called insulin resistance, which is known to be a cause of type 2 diabetes.
Co-director of the study, Chama Dibner, added: “Studies strongly suggest a link between circadian clocks, insulin resistance and diabetes development. If we establish a link between circadian mechanisms and type 2 diabetes via lipid metabolism, this could have important therapeutic implications. Thanks to our new tools for studying human muscle cellular clocks in vitro, we now have the possibility to investigate this hypothesis in our next study.”
To access the paper ‘Lipidomics reveals diurnal lipid oscillations in human skeletal muscle persisting in cellular myotubes cultured in vitro.’ published today in PNAS see http://m.pnas.org/content/early/2017/09/21/1705821114.
If you found this interesting, you might also enjoy reading:
- The Best Thing to Eat Before a Workout? Maybe Nothing at All - New York Times coverage of our research April 2016
- Eating breakfast could help obese people get more active - February 2016
- Scientists offer sweet solution to marathon fatigue - November 2015