Drug Targets, Cells and Developmental Bioscience

Uniting cell, developmental and molecular biology with pharmacology to reveal core biological processes and drive new approaches to treating health challenges.
These research themes bridge across the Department, enabling new opportunities for wider collaboration.

Uniting cell, developmental and molecular biology with pharmacology to reveal core biological processes and drive new approaches to treating health challenges.

Uniting expertise in evolution, biodiversity and conservation to explore fundamental and applied questions about life’s origins, diversity and future.

We advance infection, immunity and AMR research, exploring microbes to drive breakthroughs in public health, sustainability and the One Health agenda.

We develop and share innovative, evidence-based teaching, support professional growth, strengthen our academic community, and promote educational research.

We advance global health with innovative medicines, integrating molecular design, structural biology, and novel delivery systems for safer, effective therapies.

We advance drug delivery, precision health and medicine safety using innovative methods integrating bioinformatics and clinical data to improve equity and care.
Our research centres build a lively research community across intra- and interdisciplinary fields.

We transform innovation, arisen from basic research, to diagnostics and prognostics, medicines design and delivery, leading to personalised medicine.

We are a unique, cross-faculty research centre bridging biology, health and education.

Our mission is to improve cancer patient outcomes by fostering links between Bath’s scientists and clinicians.

From cell behaviour to ecosystem dynamics, we use mathematical modelling to help better understand living systems, making predictions and informing experiments.
99% of our submitted research activity was ranked as ‘world-leading’ or ‘internationally excellent’ in the most recent Research Excellence Framework (REF2021).

Despite big changes to their medication regimens, people being treated for drug addiction appear to be coping well in the pandemic.

A new prognostic tool developed by an international team led by CTI-Bath paves the way for personalised medicine for cancer patients.

People low on selenium are at risk of paracetamol overdose, even when they follow dosage recommendations, according to research involving Bath.

High-quality lab procedures are even more crucial to identifying effective drugs than previously thought, new research from the University of Bath reveals.

The NHS is changing the way it writes its guidelines for giving injections in hospitals, following groundbreaking research from the University of Bath.

A Bath team has been shortlisted for this year’s Emerging Technologies Competition run by the Royal Society of Chemistry.
100% of our research environment achieved a 4* or 3* rating, defined as 'world-leading' or 'internationally excellent' in the most recent Research Excellence Framework (REF2021). Learn more about how our research helps to shape a better future.
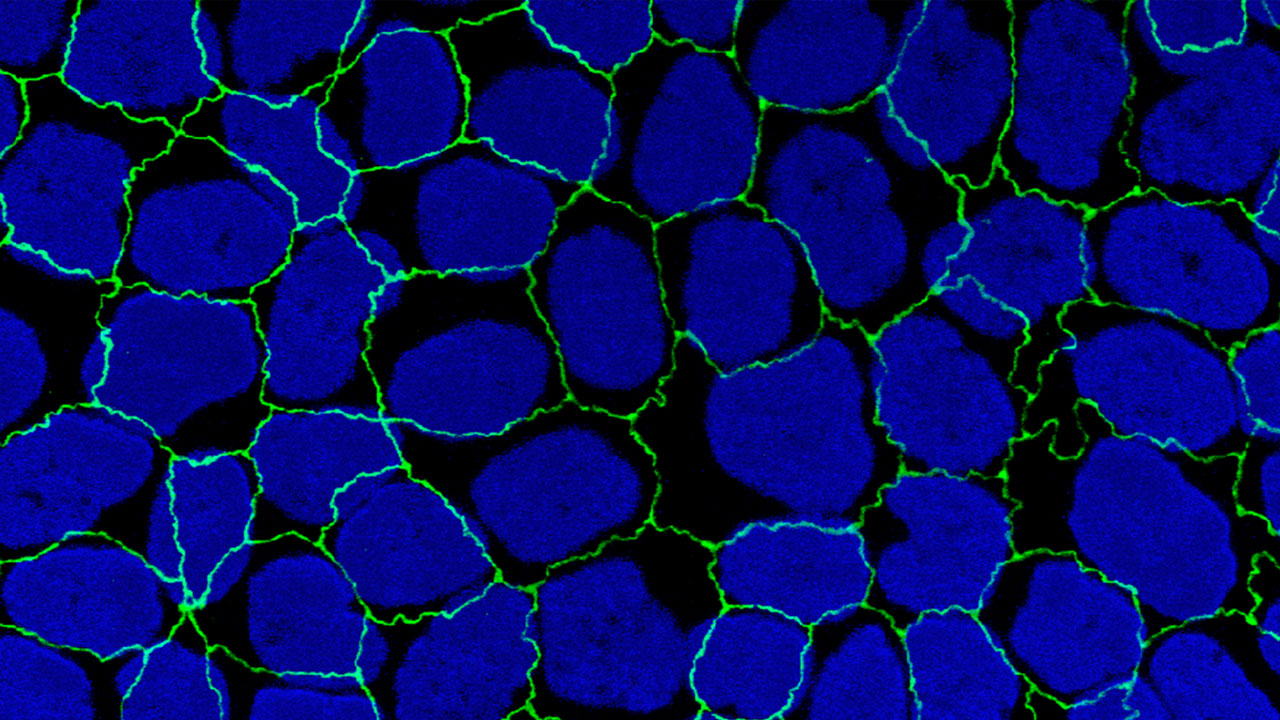
CiteAb, a new impartial citation-based search engine, is shaking up the $2 billion antibody industry.

Bath researchers collaborate with Sapience Therapeutics to find new treatments for cancer cell targets that don't respond to other drugs.
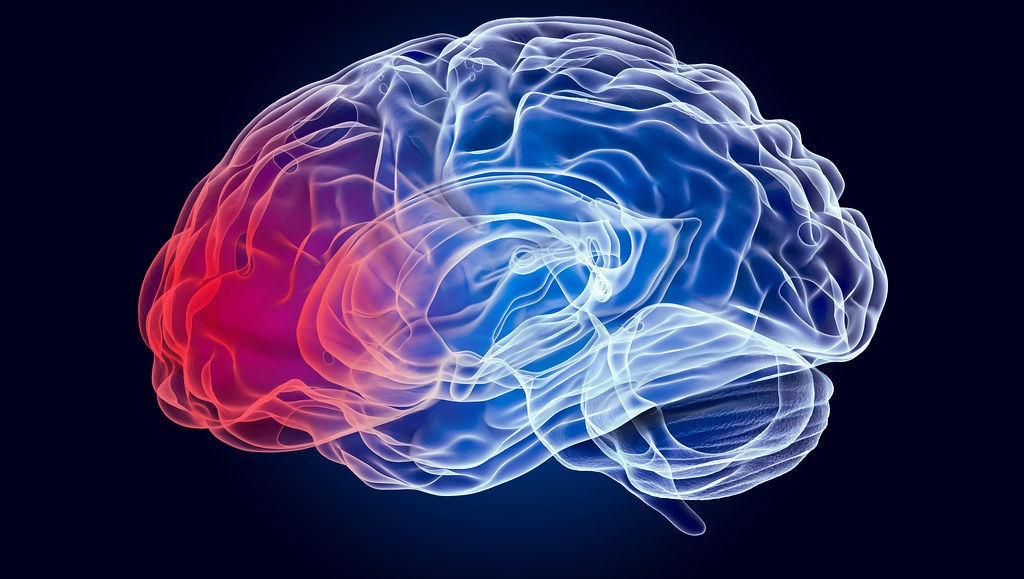
Bath scientists have discovered a series of protein structures that are thought to be highly relevant to the onset of Parkinson’s disease.

For the first time, scientists have added microscopic tracking devices into the interior of living cells, giving a peek into how development starts.

Overuse of antibiotics, high animal numbers and low genetic diversity from intensive farming increase the risk of animal pathogens transferring to humans.

Scientists at the Milner Centre for Evolution looked at the evolution of the virus that causes Covid19; their findings could help the design of a new vaccine.
Explore our state-of-the-art facilities available for teaching, research and industry with expert support from University staff.

The light microscopy network at Bath provides its users with advanced level confocal and fluorescent microscopes as well as training and advice.

A multi-user facility providing protein expression and purification services to researchers across the University and beyond.

This GW4 facility is located in Bristol and provides access to cryo-microscopy and analysis tools to study molecular processes responsible for cell function.
We offer projects over a broad spectrum of subject areas. Projects are available as part of Doctoral Training Partnerships as well as on an individual basis.

Bath hosted multidisciplinary CDT, exploring the boundaries between chemical sciences and engineering.

Delivering excellent training and research at the interface of applied mathematics, numerical analysis, probability, and statistics.

The GW4 BioMed MRC Doctoral Training Partnership aims to develop both medical and non-medical students into the next generation of medical researchers.

NERC GW4+ DTP in environmental sciences is a community of Earth, Environmental and Life Scientists.

SWBio DTP in food security & bioscience trains PhD students in the biosciences for leadership positions in academia, industry and beyond.
Find out about our PhD degrees, funding opportunities and how to apply.